All about battery safety

Batteries are an irrevocable part of vaping life. Whether it's a little cig-a-like or a massive monster mechanical mod, they all rely on a battery (or batteries) to give you those sweet clouds of craving-killer goodness.
When you start vaping you start with devices that have built-in batteries, such as the aforementioned cig-a-likes or an Ego twist. But sooner or later, you will begin to experiment with mods.
Whether a stick or box mod, these devices generally involve advanced battery technology, and by extension, owning one pays to have advanced battery knowledge. You don't need to go get a degree in electro-chemical engineering or anything, but you do need to know the basics.
So bear with me here - I'm going to assume zero knowledge of batteries, and work my way up to advanced aspects that every vaper should know, whether you plan on getting the Snow Wolf or not.
What is a battery?

A battery is a self-contained cell that stores electricity for the purpose of powering electrical devices. I'm pretty sure you know the rest.
How do batteries work?
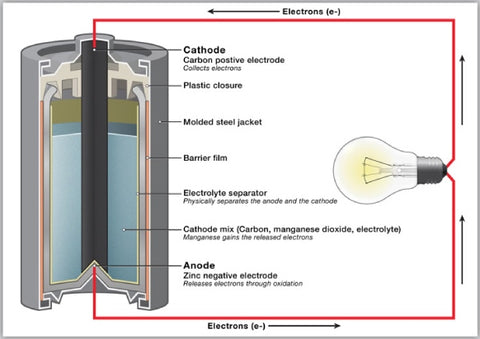
When you boil it down, all batteries work via chemical reactions.
All batteries have an anode (or negative pole), cathode (or positive pole) and an electrolyte (no, not Gatorade, a fluid or other chemical medium to act as a barrier between the anode and cathode).
The anode and cathode are made up of two different types of chemical compounds. Due to the chemical reactions within the cells, electrons build up at the anode causing a voltage difference (you may remember how voltage works from our earlier blog post about vaping and electricity).
Now electrons are fussy little beasts - they don't like being all bunched up, and would much rather spread themselves out evenly; therefore, all they want to do is go back to the cathode. But they can't, because the electrolyte is in the way. So they sit at the anode, elbowing each other for room and generally being rather keen to get moving.
When you close the circuit between the anode and cathode with a wire or other kind of conductive material, the electrons are free to go back to the cathode and finally get some personal space. As they flow along the material connecting the anode and cathode - voila - you get an electrical current.
This is how all electrically powered devices work. They draw current from one end, pass it through anything that needs electricity, then whatever is left over goes back into the battery.
Then why do batteries run out?
Remember that all batteries work via chemical reactions. As the electrons flow, it causes chemical reactions in the anode and cathode mediums that eventually cause them to stop producing electrons. This is why the voltage of a battery drops as you use it - the potential to produce a current drops as the chemical reactions progress.
So how do rechargeable batteries work?
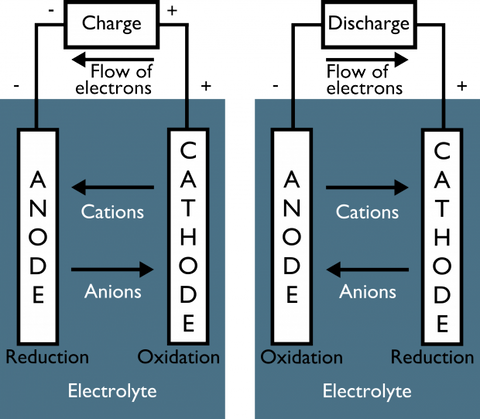
All you are doing in a rechargeable battery is reversing the flow of electrons, sending them back to the anode like trillions of microscopic four-year-olds being sent back to their rooms. The chemical reactions reverse, and the process is ready to start again.
So, those are the basics of batteries. Now on to some more interesting stuff...
Chemical makeup
Different kinds of batteries will have different chemical mediums by which they produce electrons.
Your typical car battery is a lead-acid battery. Whilst they have a low energy density (that is, capacity to store electrons), they are capable of producing massive bursts of high current - perfect for quick zaps to power your starter motor. They consist of two lead plates and sulfuric acid dissolved in the electrolyte, which is typically water.
The average battery you buy from Bunnings to power your wall clock is an alkaline battery. They actually contain manganese dioxide and powdered zinc; they get their name from the alkaline electrolyte. These are your perfect batteries for prolonged, low-current usage. For longer lasting power you can pick up a nickel cadmium battery, which were the first kind of alkaline battery.
If you owned a mobile phone in the 90s you would be familiar with the nickel-hydrogen and nickel-metal-hydride batteries. These are more energy-dense and last longer than alkaline batteries and therefore can be made to be much smaller.
Lithium-ion batteries
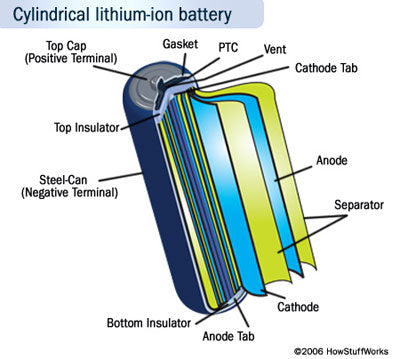
Now we're back in the present day in the world of the lithium-ion battery.
Lithium is the third element on the periodic table. It's a very small atom; likewise the least dense of all the metals with the highest electrochemical potential and energy-to-weight ratio, making it perfect for batteries. However it's also very unstable, which is why, although lithium batteries were invented back in 1912, they were not used until the development of the lithium-ion battery in the mid 80s.
Things you need to know about LiON batteries
- They are a slightly different animal to normal batteries. Lithium ions actually pass through the electrolyte to either the anode or cathode, depending on whether the battery is charging or discharging. The anode, cathode and electrolyte are actually sheets, not solid terminals in a liquid solution, so lithium-ion cells consist of the anode, cathode and electrolyte sheet wound around each other in a spiral.
- Lithium is highly reactive. The last thing you want to do is get lithium wet. Remember back to high school chemistry, when the teacher put sodium in water? Think that, but like ten times more powerful. Which is partially why...
- Lithium cells are contained under pressure. This is to keep any air or water reacting with the lithium. Therefore it is never a good idea to throw a lithium battery onto concrete as hard as you can. I mean it. Never.
So your lithium-ion battery is literally a metal canister jam-packed with a highly explosive chemical that is not unknown to occasionally catch fire! Kind of makes you think twice about having your next vape.
Fortunately for us, good LiON batteries have inbuilt safety features, which are:
- A voltage sensor. If the sensor detects that too much current is being drawn, it will restrict the flow of electrons.
- A temperature sensor. If the sensor detects that the cell is getting too hot, it will isolate the overheated part to prevent a runaway thermal reaction.
Note that I said a "good" LiON battery will have these...
Why you should be fussy about your batteries

A LiON battery will fail if you over-charge or over-discharge it.
You will over-charge it if you leave it in a charger that does not have a full charge cut off feature. Keep pumping those electrons into the battery when it's already full and the battery will let you know about it. If you're lucky it will just be ruined. If you're unlucky...
You will over-discharge it if you try to draw too much current through it. Drag 30 amps out of a 20amp-rated battery and you will degrade the electrolyte, causing the cell to overheat, and - again, if you're lucky, only ruin the battery. This is why a sound understanding of battery technology is non-negotiable for mechanical mod users - even with the battery's built-in safety features, the lack thereof in the mechanical mod can still mean you push them too hard and get yourself in trouble.
So...
What to make sure when buying high-current lithium ion batteries for your mod

- Check the ampere rating. Back in the old days this would be represented as the "C" rating, however in these more enlightened times they just write it on the side of the battery. In order to determine if the particular batteries are right for what you are going to put them through, you need to understand Ohm's law and how to calculate the amps your setup will draw. Again, because we're such legends here at Vapoureyes, we have a blog post about this.
- DON'T BUY REWRAPS! A rewrap is a battery that is not in its original skin, meaning that you can't trust the amp rating written on it. Past experience has told us that some unscrupulous characters have re-wrapped batteries with skins over-stating their amp rating, which means they can charge original price for a cheap knock-off, and you might have to suffer the consequences.
- Make sure they are protected. Make sure they have the built-in safety features mentioned above. It's as simple as making sure your vendor tells you whether they are "protected" or "unprotected". If you're highly experienced and full of expert battery knowledge (or an electro-chemical engineer), you might be able to get away with using an unprotected battery if you're very very careful with your resistances. For the rest of us - buy protected.
- Buy a decent charger. Make sure the charger you get has the full charge cut-off feature, meaning it will cut off the current as soon as it senses the batteries are fully charged.
A few other pro-tips:
- Learn Ohm's law.
- If you're using a mechanical mod, buy an ohms meter whatever you do.
- Don't leave them charging unattended - especially not overnight. You don't want to wake up with half your face blown off. You'll probably frighten yourself when you look in the mirror.
- Don't discharge them all the way. If a LiON battery gets fully discharged, it is ruined. Charge them up as soon as they dip below 20% or so.
- If you're using multiple batteries, get smart about whether to use them in series or in parallel.
- Don't drop them in water.
- Don't freeze them.
- Don't leave them in the sun.
- Don't throw them at people. It's not nice.
There's absolutely no reason to be scared of batteries. If you treat them right you will have no problems at all. But before you venture into the world of high-powered or mechanical mods, make sure you know enough to keep yourself and your loved ones safe. Batteries don't take abuse lying down!
For more information on battery and battery safety, please check out these awesome websites:
http://www.mpoweruk.com/safety.htm
http://www.livescience.com/50657-how-batteries-work.html
http://electronics.howstuffworks.com/everyday-tech/lithium-ion-battery.htm










Comments
0 Comments